Disseminating Results across Countries and Time Zones: The VOICE Trial
August 2014
Disseminating Results across Countries and Time Zones: The VOICE Trial
By Lisa Rossi, Communications and External Relations Director, Microbicide Trials Network
The VOICE trial was conducted by the Microbicide Trials Network (MTN) from 2009 to 2012 to test the safety and effectiveness of three antiretroviral-based methods of HIV prevention at 15 sites in South Africa, Uganda, and Zimbabwe.
Among those awaiting the results of the trial were the participants, members of the communities where the research was conducted, members of local community advisory boards (CABs) and institutional review boards (IRBs), government health officials, researchers, journalists, and many others working in HIV prevention around the world.
Ensuring clear, timely dissemination of study results to so many different stakeholders at multiple sites in different time zones requires careful planning and coordination. Our experience in Uganda illustrates how the MTN approached this challenge and may offer a few lessons that may be useful to other trials.
Twenty possible scenarios
The VOICE results were expected to be reported at the Conference on Retroviruses and Opportunistic Infections (CROI) in early March 2013. In our initial discussions, we identified 20 possible scenarios. We then narrowed those down to groupings of the six most likely results scenarios for each study product. The MTN was holding its annual regional meeting in Cape Town in October 2012. We took that opportunity to convene investigators, our Community Working Group, and other VOICE site team members for different group and one-on-one sessions, including a session specifically devoted to preparing for results dissemination. During that session, we drafted top-line messages for the six most likely results scenarios.
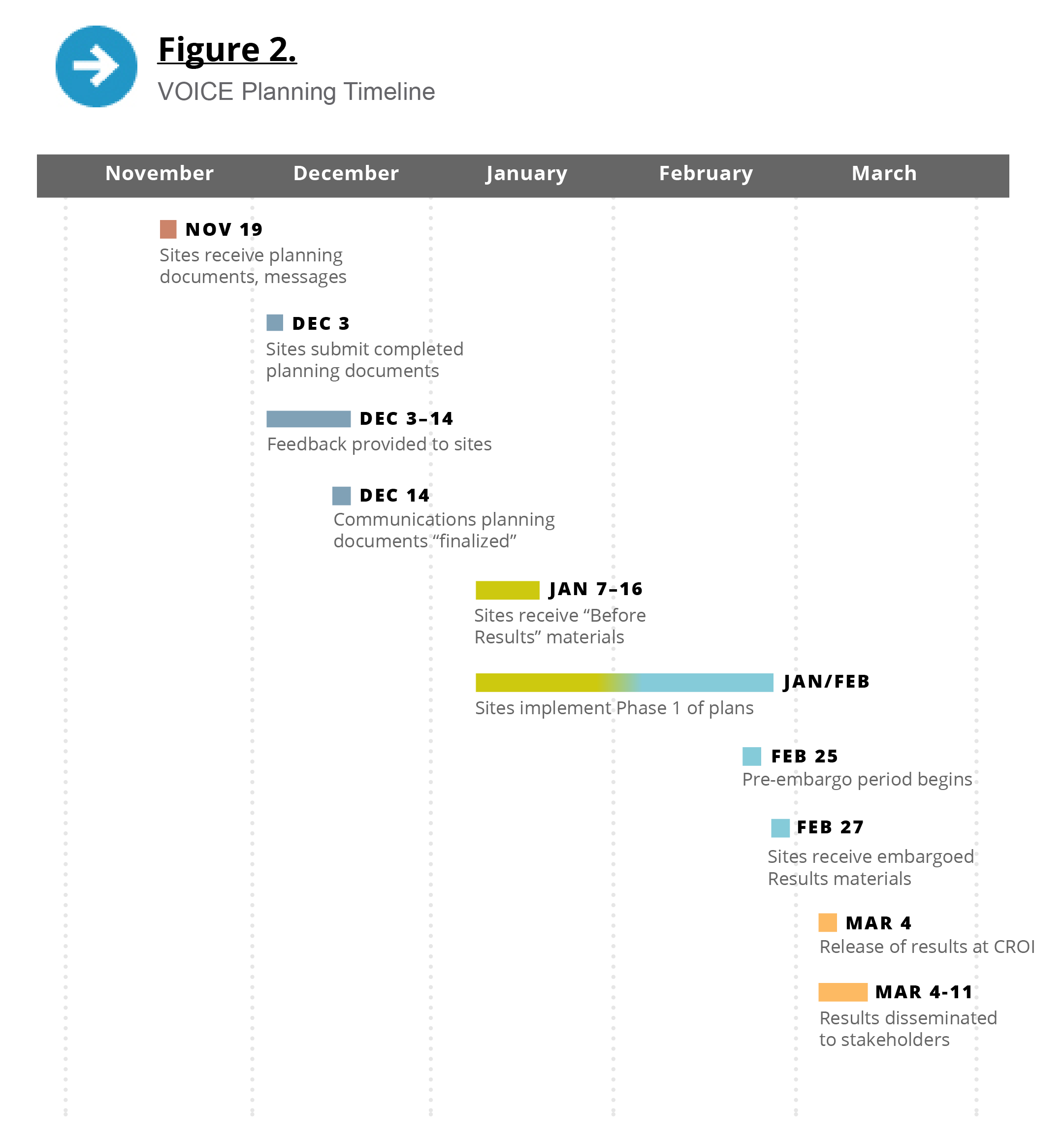
In mid-November 2012, when data analysis was nearly complete yet the results were still unknown to even the study’s lead investigators, each site received a series of communications planning documents, including materials describing the most likely results scenarios and messages for each scenario (see Figure 1). They also received the communications plan template, which I asked site staff to return by December 3.
Site staff were familiar with this document, which was similar to the templates we had used to develop the initial communications plans for the trial’s launch and the plans prepared in advance of interim reviews by the study’s data and safety monitoring board (DSMB). On two occasions, these plans were actually implemented, with the early closure of the oral tenofovir tablet arm in September 2011 and of the tenofovir gel arm in November 2011.
Like those templates, the template for the results dissemination plan began with background and a timeline of expected activities. The background section emphasized that the study’s sponsor, the US National Institute of Allergy and Infectious Diseases (NIAID), would oversee all aspects of VOICE results dissemination, including setting the parameters and the timelines for communicating results to different stakeholders. The communications team at the MTN’s Leadership and Operations Center would work with staff from each site to develop communications strategies and outreach activities within those parameters and would provide resources, guidance, and support throughout the process.
The CROI embargo
The planning template noted that because VOICE results would be reported at CROI, dissemination efforts would need to be planned around the meeting’s strict embargo policy. (Research accepted for presentation at CROI is embargoed until the time of presentation or until an official CROI press conference, whichever comes first.) There was no question of the imperative to abide by CROI’s policy. At the same time, we fully recognized our obligation to provide timely notification to participants, trial site communities and other key stakeholders, preferably before they learned of the results from other sources.
Dissemination of results would need to be highly choreographed, especially given the differences in time zones; yet, to further complicate matters, we would not know the exact date and time the embargo would be lifted until much closer to the meeting. Each day of the week and hour of the day presented a completely different set of considerations. However, for planning purposes, we assumed the embargo would end Monday, March 4, at 8 a.m. EST — 3 p.m. and 4 p.m. at the African study sites.
Throughout this time, many of us were ourselves engaged in a delicate dance. In early December, a small group of individuals was “unblinded” to the results. This group included each site’s investigator of record, but not other key site staff, many of whom were involved in the communications planning process. So, while sites were busily making plans for different scenarios, I was already preparing materials for the actual results.
First we learned the presentation would indeed take place on March 4, but we did not know what time the embargo would lift until only one week before, on February 24, when conference organizers determined that the press conference in which the VOICE results would be discussed would occur after the presentation. This meant the embargo would be lifted at the conclusion of the session — 12:15 p.m. in Atlanta and 7:15 p.m. and 8:15 p.m. in Africa. Not exactly ideal timing.
Communications planning
In November and December of 2012, the sites developed their communications plans, consulting with the MTN communications team by e-mail and occasionally by phone. My role was to guide the process and help site staff think about what they needed to consider as they developed plans tailored to their own countries and contexts.
During this time, staff at each site also updated a directory of contact information for stakeholders and the site’s standard operating procedures (SOPs) for media relations. Most of this work was done by mid-December, as we raced to get our plans and procedures in place before the inevitable lull during the holiday season in December and early January.
The site communications plans were finalized — to the extent that they could be — in December 2012. Each plan included sections representing different phases in the process: 1) communication and outreach to stakeholders before the results were reported, to discuss when to expect the results and the potential outcomes and implications; 2) pre-embargo results dissemination to selected stakeholders; and 3) public dissemination of results. They also provided information about site-specific challenges and considerations, as well as the roles of site staff who would be involved in results dissemination.
For the Makerere University-John Hopkins University (MU-JHU) clinical research site in Kampala, one consideration was the need to inform Ugandan participants in ASPIRE, another ongoing MTN trial of an antiretroviral-based HIV prevention method that had started soon after VOICE was completed The site’s plan notes that the embargo and the eight-hour time difference between Kampala and CROI’s US location in Atlanta would make it difficult to inform all the key stakeholders before they learned of the results from the media or other sources. For example, if the results were announced at CROI at 4 p.m. EST on March 4, the soonest the Ugandan site staff could talk to stakeholders about them would be the morning of March 5.
So that NIAID could determine who could be told the results in advance and when that could happen, I asked site staff to group stakeholders into three different tiers: 1) those who should be informed of the results at the earliest possible time during the pre-embargo period, 2) those who should be told as soon as possible prior to the embargo; and 3) those who should be contacted, as a courtesy, just before the lifting of the embargo.
Preparing for CROI
In January, VOICE team members at MU-JHU met with trial participants, CAB members, and journalists to inform them that the VOICE results would be announced in early March and to discuss the potential results scenarios. Other key stakeholders, including IRB members, were sent a Dear Colleague letter and materials that had been prepared by the MTN communications team, in consultation with NIAID and site staff (see Table 1). “Anticipating the Results of VOICE”— a document that provided background and context on the implications of the different scenarios and a summary of why the VOICE results would be important for various stakeholders — was particularly helpful in preparing stakeholders, notes Teopisata Nakyanzi, community engagement coordinator for MTN studies at MU-JHU.
By February 7, each site team had filled out a table specifying who would be told what, when and how, and by whom, during the final week leading up to CROI. We still did not know the date or time of the VOICE presentation or whether the embargo would be tied to a press conference, so for planning purposes we were still using March 4 at 8 a.m. EST as the end of the embargo. At the MTN annual meeting in the Washington, DC, area February 11-14, I reviewed these plans with investigators from each site. A few days later we learned the presentation would indeed be on March 4. On February 25, when I was able to notify the sites of the actual time of the embargo, I asked them to revise the tables accordingly.
On February 27 (the Wednesday before the CROI presentation), the small group of staff members who had been involved in communications planning at each site received an email with instructions on accessing embargoed VOICE materials (see Table 1) through a password-protected website.
The MU-JHU staff had planned to meet with IRB members and other Tier 1 stakeholders the week before CROI to inform them of the trial results. However, most of these stakeholders were not able to attend a meeting that week, so they were instead invited —along with trial participants to a dissemination meeting at the site on March 5.
Putting the plan in motion
The MTN communications team organized a teleconference to brief select journalists from Uganda, South Africa, and Zimbabwe at 6 a.m. EST on March 4. These journalists, and all other stakeholders who were informed of the results in advance, needed to pledge not to disclose the results until the embargo had been lifted and were warned of the consequences to the study should there be a leak. Many trial sites required signed confidentiality agreements.
When we scheduled the media briefing, we knew that we were taking a risk. But had we waited until the lifting of the embargo, it would have been too late to reach journalists in Uganda, South Africa, and Zimbabwe (8:15 p.m. in Uganda, 7:15 p.m. at the other sites), and news about VOICE results would by that time already be circulating on the Internet and wire services based on stories written by the mostly U.S. journalists attending CROI. We were careful to choose journalists who we trusted to respect the embargo and were most likely to write accurate stories. Giving them time to prepare their stories for publication immediately after the CROI session helped secure initial media coverage that was for the most part accurate, which drove subsequent coverage.
The team at MU-JHU had convened five Ugandan journalists to participate in the teleconference. In Kampala, with an eight-hour time difference, the time was 2 p.m. About 12 hours after the embargo had been lifted, on Tuesday, March 5, trial staff met at MU-JHU to hear the results. That 8:30 a.m. meeting was followed by the dissemination meeting for trial participants, members of the trial’s CAB, and IRB members and other Tier 1 stakeholders at 10 a.m.
Outreach staff had phoned the participants during the previous week to invite them to the meeting, managing to reach 300 out of 322 participants. More than half of those contacted (175) attended the meeting. Others had the opportunity to discuss the results with counselors when they came to site for an “unblinding” visit, to find out which arm of the study they had been in.
In the following days, dissemination meetings were also held with community mobilizers who had worked with the study and other stakeholders. And counselors working on the ASPIRE trial discussed the VOICE results and their implications with participants in that trial during their regular monthly visits.
The response
Despite the complexity of both the dissemination effort and the messages about the trial, everything went smoothly. The media coverage was generally accurate and balanced, causing no misunderstandings. Stakeholders had questions about the results, but they were able to get answers from the investigators and other trial staff.
Community Coordinator Nakyanzi admits that she and others at MU-JHU had been anxious about the potential response to the VOICE results. “But the stakeholders were actually very supportive,” she said.
Nakyanzi believes that all of the work that went into preparing the study teams and stakeholders for results dissemination minimized misrepresentation of the results and negative public reaction. “When the participants clearly understand the outcome, they share what they think could have happened, and there is an opportunity for feedback, participant counseling, and future steps,” she said.
Our story did not receive a great deal of media coverage. It was overshadowed by the big news at CROI 2013, about the “Mississippi baby,” a child who appeared to have been “cured” of HIV. But our objective was not to garner extensive media coverage. Given the results we had to share — that none of the study products proved effective and most participants did not use them daily as directed —our priority was ensuring that news reports were accurate. This meant getting the facts right, but also acknowledging that the study had answered the question it set out to address, rather than spreading the misperception that the study had “failed.” Our monitoring of the coverage found that these efforts were successful.
Lessons learned
Planning pays off. Even before we knew exactly when the results could be made public, our plan was in place. It spelled out exactly what would be done and by whom, and later we were able to add specific dates and times. Key messages had been developed for each of the most likely results scenarios, giving us a head start on materials development. As a result, “we were able to use the planning and background documents to contact different groups at different times and complete dissemination activities in a timely manner, preventing possible misinformation,” says Nakyanzi.
Ongoing stakeholder engagement is important. All of the trial sites had been engaging with participants, community members, health officials, local leaders, journalists, and other stakeholders throughout the trial. Results dissemination was not the first time that stakeholders had heard from VOICE. Study staff had established themselves as trustworthy sources of information, and they had experience with communicating study outcomes and the research process to a range of audiences.
Preparation lays a strong foundation for results dissemination. Site staff were familiar with the communications planning documents and the process, which began at the start of the VOICE trial. They had used earlier versions of the templates to help create the overall VOICE communications plan and prepare for the closing of two arms of the trial. They also had SOPs and stakeholder directories, which they just needed to update. Throughout the trial, we had done scenario planning and messaging in consultation with site staff, so everyone was knowledgeable about the final key messages. All the investigators had received training in media communication at the beginning of the trial, and we were able to provide additional one-on-one media training at the MTN Regional Meeting in October.
Managing expectations is important. It is important to prepare not only staff but also other stakeholders for potential outcomes, so that no matter the results, they can be accepted for what they are. Ongoing communication about the research process with all of our stakeholders, including those involved in other HIV prevention trials, helped them put the VOICE results into perspective.