How Social Media Affects Results Dissemination: HVTN 505
July 2014
How Social Media Affects Results Dissemination: HVTN 505
By Jim Maynard, HIV Vaccine Trials Network, Director of Communications and Community Engagement, Seattle, WA, USA
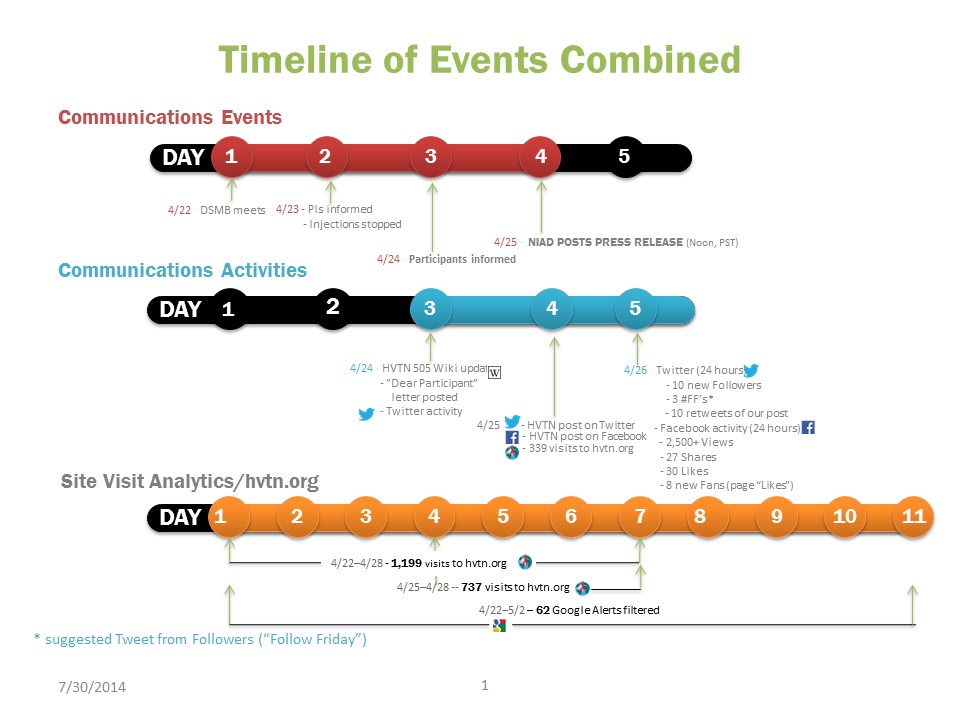
When the Data Safety Monitoring Board (DSMB) for the HIV vaccine efficacy trial known as HVTN 505 met on April 22, 2013, for a planned interim review of the study data, a comprehensive communications plan was already in place.
The communications teams from the US National Institute of Allergy and Infectious Diseases (NIAID) and the HIV Vaccine Trials Network (HVTN), in collaboration with the HVTN 505 Protocol Team leadership, NIAID’s Vaccine Research Center representatives, and consultants from the firm SHI Consulting, had developed a plan that addressed a number of possible scenarios. Therefore, we were prepared when the DSMB recommended that study injections be stopped because the data showed that the vaccine regimen under investigation was not efficacious.
When a clinical trial undergoes any major changes, or when there are important updates to share, our first responsibility is to make sure the trial participants are briefed and have a chance to ask questions before the information is made public. Our goal is for the participants to hear the information from a study clinician whom they know and trust, rather than on the nightly news broadcast or in the morning newspaper. Our plan included briefing the principal investigators at all trial sites and giving them time to contact their participants and/or bring them into the clinic.
What we did not foresee was that social media would become the primary channel for the dissemination of these HVTN 505 interim results. Our experience holds important lessons for other trials as they develop communications plans, seeking to strike the ideal balance between social and traditional media.
The day of the DSMB meeting, the communications plan went into effect. On the night of April 22, the principal investigators at each of the 21 study sites in 19 US cities were informed of the results, and the sites stopped giving study injections on April 23. Study staff began contacting participants as early as the evening of April 22, following the briefing with principal investigators.
A total of 2,504 men and transgender women had volunteered to receive four injections of the study vaccine regimen over a six-month period. We were determined to ensure that they heard the news from study staff before they heard it through the grapevine, so we knew we needed to move fast.
NIAID planned to send out a news release and a question-and-answer document about the trial late in the day on April 25, thus allowing the sites sufficient time to inform the participants first. By the end of the day on April 24, most participants had been reached. But in a world where many of those participants are social media savvy and “plugged in” 24/7, as soon as you’ve told one participant, the race is on.
On April 24, prior to the NIAID press release, one of the participants posted the “Dear Participant” letter on his blog, which others reposted and tweeted. (This letter to participants had been developed as part of the communications plan and subsequently updated with more specific information after the DSMB review.) Someone also updated the HVTN 505 page on Wikipedia, though less accurately.
One participant, a blogger with an active site and thousands of followers, blogged about the stoppage of injections in HVTN 505. A reporter for Nashville’s Tennessean saw the blog and interviewed the participant for a story that appeared in the local print media.
The story was out. NIAID sent out its news release earlier than planned, in the early afternoon of April 25. Once the news release was available, the HVTN team posted to Facebook and tweeted about the study closure as planned.
Within 24 hours, the HVTN Facebook page had received more than 2,500 views and gained eight new fans or page “likes.” The Facebook post with the NIAID press release was shared 27 times and was “liked” 30 times. On Twitter, the HVTN’s tweet was retweeted 10 times and HVTN gained 10 new followers. Many of the HVTN’s clinical trial sites used the posts from the HVTN on their sites’ Facebook pages and Twitter feeds.
The activity on social media was heaviest during the 24 hours after the NIAID news release was posted, but it continued for several days, helping drive traffic to information about the trial on the HVTN website.
From April 22 to 28, www.hvtn.org received 1,999 visits, an increase of over 600 visits compared to 1,359 in the same period the year before. Google alerts on media coverage of the trial showed that 62 articles were published from April 22 to May 2.
Did the “leak” of the story through social media hurt our dissemination efforts? Fortunately not. Because the original blogger and other participants relied on an accurate source of information – the Dear Participant letter – their efforts reinforced the official dissemination of the trial’s interim results. Likewise, the first news article published about the changes to the trial was excellent and accurate.
So, this case study is not a cautionary tale about the perils of communicating HIV prevention trials information in the age of social media. It is, however, a reminder that we must consider the growing power and reach of social media in many communities as we develop communications plans for clinical trial, and that study sponsors are not the only messengers.
In the case of HVTN 505, the potential for negative consequences because of a leak was very real. The halting of study injections was an emotional topic for participants, and in the early days after the DSMB meeting there was a concern, which was unfounded, that the investigational vaccine regimen had increased the risk of HIV infection among participants who received it. It would have been easy to get the story wrong, but almost no one did.
As platforms such as Faceboook and Twitter grow in popularity in many countries throughout the world, the HVTN 505 experience shows just how important it is to account for social media in communications planning to avoid the potential pitfalls and capitalize on its advantages. If trial participants are active on social media, the trial’s communications team must move quickly to synchronize communication about results via traditional and social media with participants’ own efforts to share the news with their personal social networks.
To suggest additional case studies, email commshandbook@avac.org.